|
Home, Search, Index, Links, Pathology, Molecules, Syndromes, Muscle, NMJ, Nerve, Spinal, Ataxia, Antibody & Biopsy, Patient Info |
HEREDITARY SENSORY & AUTONOMIC NEUROPATHIES (HSAN)

|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Polyneuropathy with Minifascicles, 46,XY Gonadal dysgenesis & Mental retardation (GDMN)
● desert hedgehog (DHH)
|
|
Posterior column ataxia & Retinitis pigmentosa (AXPC1; PCARP)
● Feline leukemia virus subgroup C cellular receptor 1 (FLVCR1)
- Epidemiology: Multiple families
- Genetics
- Mutations: > 20 described; Asn121Asp, Cys192Arg, Ala241Thr
- Allelic disorder: HSAN
- Diamond-Blackfan anemia

- Enhanced alternative FLVCR1 splicing
- No FLVCR1 mutations found
- FLVCR1
- High expression in retina, spinal cord & cerebellum
- MFS transporter
- Single-polypeptide secondary carriers
- Transport small solutes in response to chemiosmotic ion gradients
- Exports cytoplasmic heme in erythrocyte precursors
- Possible disease mechanism
- Perturbation of neuronal heme transmembrane export
- Abrogates neuroprotective effects of neuroglobin (NGB)

- Initiates apoptotic cascade
- Other iron homeostasis disorder: Friedreich ataxia
- Clinical
- Onset: Early childhood; Visual loss
- Visual
- Ring scotoma
- Nyctalopia (Night blindness)
- Progessive loss: Loss of central vision; To blindness
- Retinitis pigmentosa: Pan-retinal loss of pigment epithelium
- Sensory
- Onset: 2nd decade
- Loss: Large fiber; Position sense; Apallesthesia
- Pain & Temperature: Preserved
- Sensory ataxia
- Progression to severe disability
- Tendon reflexes: Absent
- Autonomic
- GI: Achalasia; Dysmotility
- GU: Incontinence in some patients
- Skeletal: Scoliosis; Camptodactyly
- Laboratory
- Electrodiagnostic: Sensory ganglionopathy; Absent SNAPs
- MRI: Posterior column abnormality
- Pathology
- Peripheral nerve: Loss of large myelinated axons
- Muscle: Normal
- Dorsal root ganglia: Neuron loss
- CNS: Posterior column damage
- FVLCR1: Variant: HSAN
- Epidemiology: 7 patients
- Genetics
- Inheritance: Recessive
- Mutations: Missense
- Clinical
- Onset age: Early childhood
- Pain insensitivity
- Autonomic: Hypohidrosis
- Tendon reflexes: Reduced or Absent
- Extremity mutilation
- Strength: Distal arm & leg weakness in some patients
- Retinitis pigmentosa
- Developmental delay
- Laboratory
- Nerve pathology: Axon loss
- NCV: Axon loss
- Muscle pathology: Type grouping; Grouped atrophy
- Brain MRI: Delayed myelination
- PCARP phenotype without mutations
- Additional feature: Loss of pain sensation on hands & feet
Hereditary Sensory Neuropathy with Gastro-Esophageal reflux & Cough (HSAN IB)
● Chromosome 3p24-p22; Dominant
- Clinical
- Onset
- Age: 2nd to 5th decade
- Cough or Gastro-Esophageal reflux
- Cough
- Associated with
- GE reflux
- Impaired laryngeal sensation (Denervation hypersensitivity)
- Triggered by
- Noxious odors
- Pressure in external auditory canal (Arnold's ear-cough reflex)
- May produce syncope
- Associated with
- Gastro-Esophageal reflux
- Clinical: Heartburn; Regurgitation
- pH < 4 in esophagus
- Onset precedes neuropathy
- Pharyngeal, other: Some due to reflux
- Throat clearing
- Hoarse voice
- Neuropathy
- Onset: 3rd to 5th decade
- Sensory loss
- Distal: Some patients also with truncal involvement
- Legs & Arms
- Pain & Temperature: More severe than large fiber modality involvement
- Painless injuries: Common
- Lancinating pains: Occasional
- Ulcers unusual
- Other: Some patients
- Autonomic: Impotence; Alacrima; Hypohidrosis, distal
- Hearing loss: Sensorineural
- Retinal detachment
- Onset
- Laboratory
- Nerve conduction: Sensory, axonal loss
- Audiometry: Sensorineural hearing loss (40%)
- Nerve pathology: Loss of large & small axons
Hereditary Sensory Neuropathy with Deafness and Global Delay 2
● Autosomal Recessive
- Epidemiology: Lebanese Moslem family
- Onset
- 2 months to 3.5 years
- Developmental delay: Motor; Language
- Clinical
- Developmental delay (100%): Motor; Language
- Hypotonia (100%)
- Areflexia (100%)
- Hearing loss (100%): Sensorineural
- Ataxia (75%)
- Weakness (50%): Proximal
- Dysmorphic features (50%)
- Other: Optic atrophy; Renal tubular acidosis
- Electrophysiology: Neuropathy
- Sensory
- Axonal loss
- Sural nerve biopsy: Axonal loss
Hereditary Sensory Neuropathy with Deafness & Dementia (HSN 1E)
● DNA methyltransferase 1 (DNMT1)
- Epidemiology: > 33 families
- Genetics
- Mutations
- Missense: C353F; T481P, Tyr495Cys; Asp490Glu–Pro491Tyr; P491L; Y524D; I531N; His569Arg
- Location
- Target sequence domain: Middle or N-terminus
- Exons 20 & 21
- Common mutation: Tyr495Cys
- Allelic disorders
- Autosomal Dominant Cerebellar Ataxia, Deafness & Narcolepsy (ADCA-DN)
- Mutation location: C-terminus of Target sequence domain; Exon 21
- Spastic paraplegia
- Mutation: c.2053G>A, p.A685T
- Inheritance: Dominant
- Autosomal Dominant Cerebellar Ataxia, Deafness & Narcolepsy (ADCA-DN)
- Mutations
- DNMT1 protein
- Components of DNA replication machinery during S phase of cell cycle
- Maintenance of methylation, gene regulation & chromatin stability
- Transfers methyl groups to cytosine nucleotides
- Maintains methylation patterns following DNA replication
- Mediates transcriptional repression: Direct binding to HDAC2
- Immune cells
- Highly expressed
- Required for differentiation of CD4+ into T regulatory cells
- Mutant proteins: Reduced enzymatic activity & heterochromatin binding
- Other disorders of DNA methylation related genes
- Clinical
- Onset
- Age: 16 to 51 years; Mean 38 years
- Hearing loss: Most common
- Hearing loss
- Often at onset
- Sensorineural
- May be treated with Cochlear implant
- Sensory
- Loss
- Extremities
- Distal > Proximal
- Legs > Arms
- Large & Small axon modalities: Vibration > Joint position
- Lancinating pain: Some patients
- Loss
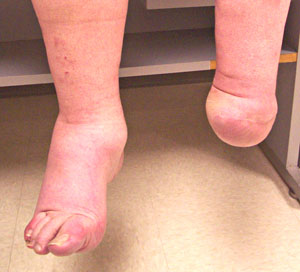
- Ulcero-Mutilation
- Neuroarthropathy
- Painless fractures
- Tendon reflexes: Reduced distal
- Weakness: 15%; Distal
- CNS
- Ataxia: 15%
- Gait: Unstable
- EOM: Square wave jerks
- Dementia: Cognitive decline
- Onset 4th & 5th decade
- Personality changes & Psychiatric manifestations
- Frontal lobe dysfunction
- Clouded consciousness
- Sleep disorders
- Narcolepsy
- Hypersomnia
- Rapid eye movement sleep disorder
- Seizures: Myoclonic
- Hallucinations: Auditory & Visual
- Ataxia: 15%
- Systemic: Renal failure
- Death: 5th & 6th decade; Mean 54 years
- Onset
- Laboratory
- EEG: Low frequency (delta waves), frontal-predominant
- Electrodiagnostic
- SNAPs: Often absent
- Motor: NCV normal or slightly slow; CMAP normal
- Brain MRI: Atrophy
- Pathology
- Posterior column: Especially medial
- Cerebellum: Purkinje cell swelling & axonal loss; Hyperplasia of Bergman glia
- Inferior olivary nucleus: Neuron loss
- Cerebral atrophy
- Nerve: Axon loss, Severe, Large & small
- Mutant DNMT1: Translocate to cytoplasm: May form aggresomes
● AIFM1; Chromosome Xq26.1; Recessive
- Epidemiology: Chinese family
- Genetics
- Clinical
- Onset
- Age: Mean 13 years
- Hearing loss: Low frequency
- Hearing loss: Auditory neuropathy
- Early: Low frequency
- Progression: To all frequencies
- Poor speech discrimination
- Absent BAERs: Early
- Intact outer hair cell function
- Neuropathy: Sensory
- Onset
- 3rd or 4th decade
- After hearing loss
- Sensory loss
- SNAPs: Absent or Reduced amplitude
- Sensory NCV: Mildly reduced
- Motor NCV & CMAPs: Normal
- Onset
- Onset
Gyrate atrophy of choroid & retina with hyperornithinemia (GACR)
● Ornithine aminotransferase (OAT)
- Genetics
- Mutations: Misense & Stop
- OAT protein
- Catalyzes reversible transamination of ornithine to glutamate semialdehyde
- Clinical
- Chorioretinal degeneration
- Cataract formation: Early
- Laboratory
- Hyperornithinemia; No hyperammonemia or homocitrullinuria
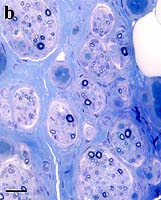
- Muscle: Type II fiber atrophy; Tubular aggregates
- Nerve: Sensory axonal neuropathy, by electrophysiology
Hereditary Sensory & Autonomic neuropathy (HSAN) with Anosmia
● ? Autosomal Recessive
- Epidemiology: 1 Japanese family
- Onset
- Age: 20-50 years
- Anosmia
- Special sensory
- Anosmia: All 3 family members
- Ageusia: 1 person
- Autonomic
- Anhidrosis: Diffuse
- Orthostatic hypotension
- Sensory loss
- Large & Small fiber modalities
- Distal
- No skin ulceration
- Electrodiagnostic
- Motor conductions: Normal
- Sural responses: Absent
- NCV: Normal
- Skin biopsy: Eccrine gland - vacuolar degeneration & dilated ducts
- Sural nerve: Reduced number of myelinated & unmyelinated axons
Hereditary sensory & autonomic neuropathy type IID (HSAN2D)
● SCN 9A
- Epidemiology: Japanese patients
- Genetics
- Mutation: c.3993delGinsTT; Premature stop
- Allelic disorders
- SCN 9A protein: See SCN 9A & Erythromelalgia
- Clinical
- Onset age: Congenital to 3rd decade
- Sensory
- Pain & temperature loss: Progressive; Distal > Proximal
- Vibration sense: Preserved
- Autonomic
- Hypohidrosis
- Urinary bladder dysfunction
- Lacrimation: Reduced in 1 patient
- Tendon reflexes: Normal, Increased or Reduced
- Strength: Normal or Reduced in legs
- Hearing loss: Asymmetric
- Hyposmia
- Hypogeusia: Fungiform papillae on tongue reduced
- Skin: Painless ulcers; Rash
- Skeletal
- Recurrent fractures
- Short digits
- Bone dysplasia
- Laboratory
- Sensory nerve conductions: SNAP amplitudes mildly small or Normal; Asymmetric
- Sural nerve: Loss of large myelinated axons or Normal
Congenital inability to experience pain (CIP)
● SCN 9A
- Genetics
- Mutations: Nonsense (S459X, I767X, W897X); Splicing; Missense (Arg896Gln)
- CIP also described in mixed breed dogs 19: Arg921Cys homozygous mutation
- Allelic disorders
- SCN 9A protein: See SCN 9A & Erythromelalgia
- Clinical
- Onset age: Congenital
- Sensory
- Inability to feel pain
- Over all body regions
- Painless injuries present
- Some patients: Hyposmia; Hypogeusia
- Normal: Touch; Temperature (warm & cold); Proprioception; Tickle; Pressure
- Inability to feel pain
- Motor: Normal strength
- Tendon reflexes: Normal
- Sweating: Present
- Bladder function: Normal
- Laboratory
- Nerve conduction studies: Normal
- Nerve biopsy: Normal
- Skin pathology: Loss of Meissner’s corpuscles & Epidermal nociceptors
- CNS MRI: Normal
- Differential diagnosis
Paroxysmal Extreme Pain Disorder (PEPD; FEPS4)
● SCN 9A
- Nosology: Familial rectal pain; Familial Episodic Pain Syndrome 4 (FEPS4)
- Genetics
- Mutations: Missense; Present in 2/3 of families with PEPD
- Allelic disorders
- SCN 9A protein: See SCN 9A & Erythromelalgia
- Mutated protein: Altered inactivation with associated persistent currents
- Onset: Congenital
- Clinical
- Paroxysmal
- Pain
- Severe
- Burning
- Locations: Rectal, Genetalia, Face (Ocular, Mandibular)
- Associated with: Non-epileptic tonic seizures; Cardiac asystole
- Treatment: Carbamazepine
- Autonomic dysfunction
- Flushing of skin: Harlequin colour changes
- Pupillary abnormalities
Polyneuropathy & Erythromelalgia 16
● Nicotinamide Nucleotide Adenylyltransferase 2 (NMNAT2)
- Epidemiology: 2 families, 4 patients
- Genetics
- Mutations: Missense; T94M, V98M, R232Q
- Allelic disorders
- Fetal akinesia deformation sequence
- Mouse model: Motor neuropathy
- NMNAT2 protein
- Clinical
- Onset ages: 1 to 4 years
- Pain: Feet
- Episodes
- Stimuli: Exercise, Heat, Infections, Stress
- Duration: 1 to 30 days
- Frequency: Every 2 to 3 months
- Foot pain & swelling
- Weakness: Some patients; Proximal, Distal, Respiratory
- Course: Progressive distal weakness + Episodes
- Skeletal: Pes cavus & equinus; Scoliosis
- Laboratory
- Nerve biopsy: Axon loss & regeneration
- Electrodiagnostic: CMAP ± SNAP amplitudes reduced; NCV normal
- MRI: Normal brain & spinal cord
- NMNAT2 variant syndrome: Fetal akinesia deformation sequence
17
- Epidemiology: 1 family, 2 patients
- Genetics
- Inheritance: Recessive
- Mutations: Loss of function; c.403dupC, c.695G>A (p.R232Q)
- Clinical
- Onset age: Prenatal
- Cystic hygroma
- Skin edema
- Ascites
- Pleural effusion
- Hydrocephalus
- Muscle: Small; Replaced by fat
- Contractures: All extremities
- Laboratory
- CNS: Small; Hydrocephalus
CMT 2V: Hereditary Adult-onset Painful Axonal Polyneuropathy
● α-N-acetyl-glucosaminidase (NAGLU)
- Epidemiology: French-Canadian family & North American
- Genetics
- NAGLU protein
- Lysosomal
- Degradation of heparan sulfate (glycosaminoglycan compound)
- Mutation: 45% reduced enzyme activity
- Immune disorder of heparan sulfate component: Neuropathy with IgM vs TS-HDS
- Clinical
- Onset age (Pain): 12 to 61 years
- Sensory
- Pain: Legs; Sleep disorder (Cramps or Paresthesias)
- Ataxia: Reduced tandem walking; With disease progression
- Loss (50%): Pain & Vibration
- Tendon reflexes: Normal of Reduced
- Strength: Normal
- Laboratory
- NCV: SNAP & CMAP amplitudes normal or reduced with disease progression
- EMG: Usually normal
Sensory (Small fiber) Neuropathy + Deafness-Dystonia & Ichthyosis 15
● Fat-Inducing Transcript 2 (FITM2; FIT2)
- Epidemiology: Pakistani family, 5 patients
- Genetics
- FITM2 protein
- Family: Involved in partitioning of triglycerides into cellular lipid droplets
- Neutral lipid storage & metabolism
- Lipid droplets
- Organelles that store neutral lipids
- Cytoplasmic
- Surface: Phospholipid monolayer
- Modulate cellular signaling, lipid metabolism, transcriptional regulation, autophagy & immunity
- Lipid droplet-ER contact
- May be depleted by: Tamoxifen
- Disorders of proteins involved in lipid droplet biogenesis
- BSCL2 (Seipin): Berardinelli–Seip congenital lipodystrophy; SPG 17; HMN 5C
- Tamoxifen enteropathy
- Clinical
- Onset age: 6 months
- Sensorineural hearing impairment: Earliest feature: Progression to severe deafness
- Global developmental delay
- Neuro-regression
- Walking: Late or None
- Failure to thrive: Low weight
- Speech reduced
- Dystonia: Hands
- Contractures: Pes cavus
- Muscle wasting: Legs
- Sensory abnormalities
- Not length dependent
- Pain: Joints; Paresthesias or burning in distal linbsa, joints & trunk
- Pain sensation: Loss in arms & face; Preserved in trunk &legs
- Ichthyosis: Especially shin & scarring alopecia
- No lipodystrophy
- Laboratory
- Serum CK & aldolase: Normal
- NCV: CMAPs normal; SNAPs normal
- EMG: Normal
- Brainstem-evoked response audiometry: No responses
- Brain MRI: Normal
- Muscle pathology: Normal
Hereditary Sensory ± Motor Neuropathy 20
● INSC Spindle orientation adaptor protein (INSC)
- Epidemiology: Asian family; 8 patients
- Genetics
- Mutation: Missense; Met70Arg
- INSC protein
- PAR3(PARD3)/INSC/LGN(GPSM2) complex
- Cell division, asymmetric
- Mutation effect
- Tubulin aggregation: Reversed by microtubule stabilization
- Haploinsufficiency
- Clinical
- Onset age: 7 to 29 years
- Sensory loss: Large fiber or Pan-modal; Distal
- Paresthesias (40%): Hands & Feet
- Weakness (40%): Distal; More after 50 years of age
- Gait disorder
- Skeletal: Pes cavus
- Course: Slow progression
- Laboratory
- NCV
- Motor: Conduction velocities 48 to 63 M/s; CMAP ampltudes normal to mildly reduced
- Sensory: SNAPs often absent
- NCV
Animal model of HSN: mutilated foot (mf) rat 5
● Chaperonin containing T-complex polypeptide 1, subunit 4 (CCT4)
- Genetics
- Missense mutation: Tyr450Cys
- Chromosome: Syntenic to human 2p13–p16
- No human mutations yet identified
- CCT4 protein
- Involved in folding newly translated peptides: Tubulin, actin, α-transducin & other cytosolic proteins
- Mechanism: Multiple rounds of ATP-driven release and rebinding of partially folded intermediate forms
- Clinical features
- Ataxia
- Insensitivity to pain
- Foot ulceration
- Pathology
- Severe loss of sensory ganglia & axons
- Apoptosis of cells during development
Congenital Indifference to Pain 7
- Definition: Loss of pain sensation with normal peripheral nerve function & anatomy
- Epidemiology: Individual case reports
- Onset: Congenital
- Clinical
- Sense of pain to noxious stimulis: Reduced or Absent
- Temperature: Perception of hot & cold reduced
- Itch: Reduced or Absent
- Taste: Normal except reduced perception of spicy foods
- Normal: Light touch; Proprioception; Vibration; Autonomic
- Pharmacology: Mild improvement in pain perception with naltrexone
- Laboratory
- EMG & NCV: Normal
- Pathology: Normal anatomy of PNS & CNS sensory pathways
- Differential diagnosis: CNS lesions
- Asymbolia for pain
- Lesions in cingulate, insular, or somatosensory cortical regions
- Reduced affective-motivational component of pain perception
- Right postcentral gyrus lesion
- Inability to properly discriminate pain stimuli
- Retained capacity to react appropriately to pain
- Psychitric disorders
- Asymbolia for pain
Return to Neuropathy differential diagnosis
Go to Neuromuscular home page
References
1. Am J Hum Genet 2000;67, Acta Neuropath 1999;98:309-312, Ann Clin Transl Neurol. 2017;4:415-421, J Peripher Nerv Syst 2020;25:423-428
2. Pediat Neurol 2002;27:49-52
3. Arch Neurol 2003;60:329-334
4. Am J Hum Genet 2003;Online July
5. Hum Molec Genet 2003;12:1917-1925
6. Acta Neurol Scand 2001;104:316-319
7. Pain 2006;122:210-215
8. J Med Genet 2006;43:e33
9. Nature 2006;444:894-898
10. Neuron 2006;52:767–774; Journal of Neurology, Neurosurgery, and Psychiatry 2006;77:1294-1295
11. Am J Human Genet 2010;87:643–654
12. Nat Genet 2011 May 1, Brain 2015; Online Feb, J Peripher Nerv Syst 2023 May 18
13. Neurology 2013;80: Online April
14. Brain 2015; Online March
15. Dis Model Mech 2017;10:105-118
16. Exp Neurol 2019 May 24, J Clin Invest 2022 Oct 26;e159800
17. Exp Neurol 2019 May 25
18. Eur J Med Genet 2020;63:104037
19. J Vet Intern Med 2023 Jan 11
20. EMBO Mol Med 2024 Apr 8
4/10/2024