Possible severe side effects
- Encephalopathy
- Bone marrow suppression: Anemia, Thrombocytopenia Neutropenia
Admission testing
- Patient is admitted for treatment by staff.
- On admission
- Vital signs: Obtain on initial evaluation and Hourly during infusion
- Height and weight: Measure for Fludarabine dose calculation.
- Patient is evaluated by a study physician: Obtain
- Interval history
- Pertinent physical exam
- Adverse events
- Quantitative dynamometry: On first and last visit; Performed by Dr Pestronk
- Laboratory
- Blood: CBC and electrolytes, glucose, and creatinine (BMP)
- First and last admissions: Blood is also drawn for
- Antibody titers to GM1 ganglioside or MAG: 2 red top tubes to Neuromuscular Lab
- Immunocompetency panel: Send for CD3, CD19
- Quantitative immunoglobulins
IV Access
- Start IV heparin lock
- Start normal saline at 100 cc/hr
- Run saline at keep vein open (kvo) rate during infusion
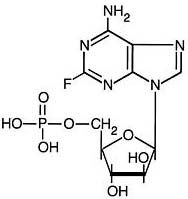
Fludarabine Infusions: 5 consecutive days each month x 6 months
- Fludarabine dose: 25 mg/M2. Calculate absolute dose to be administered.
- Fludarabine Infusion: Over 30 minutes
- Patients may take all regular medicines while in hospital.
Precautions
- Problems: Contact the Neuromuscular fellow immediately.
- A fellow is available 24 hours a day
- Phone #: 388-6138
- Notify fellow for
- Temp > 38.0
- Cardiac complaints
- Respiratory symptoms: Shortness of breath must be reported immediately.
- Encephalopathy: Mental status change; Visual loss
- Rash
- Activity while in hospital: Patient may be up ad lib
Discharge
- Patient should be sent home on their regular medicines
- Follow-up: 2 months after final infusion in the Neuromuscular clinic.
- Questions or problems: Patient should call Neuromuscular office at 314-362-6981.
|
|
| Fludarabine |
|---|
|